Clinical Outcomes and
Applied Research Studies
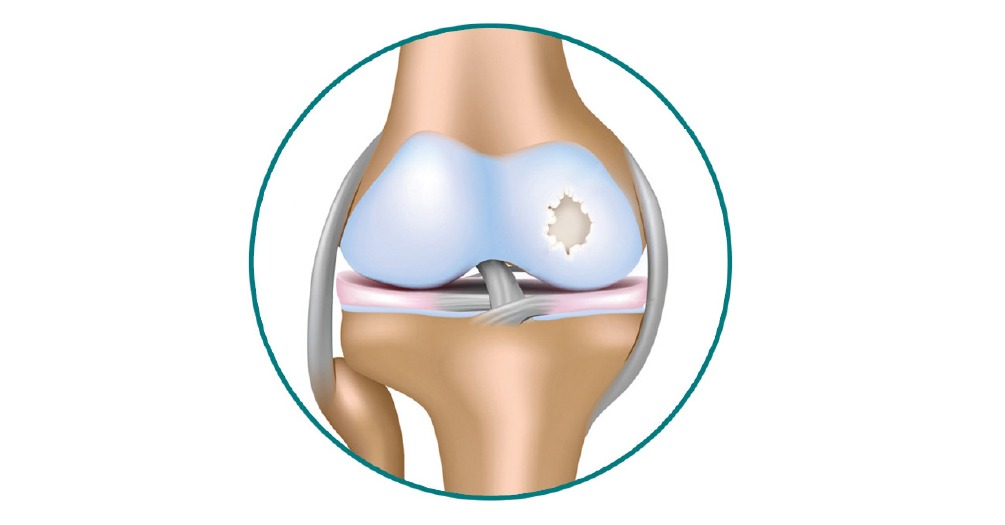
Orthobiologics refers to the treatment of musculoskeletal disorders using different autologous and allogenic products. This treatment modality has been incorrectly advertised as “Stem Cell Therapy” however importantly these products often do not contain any stem cells or in such a small amount to not have patient benefits. Further there is no scientific evidence of any regenerative or direct reparative effect of any of these products despite the majority of advertising on the internet and clinics stating the opposite. Recently Kingery et al (JBJS 2020) reported that 95.9% of practice websites contained at least one statement of misinformation and “failed to accurately represent the clinical efficacy” of “Stem Cell Therapy”. It is in fact unethical and illegal to advertise or promote such deceptive claims and accordingly physicians and hospital systems are currently experiencing great difficulty in responding to patients requests for these types of treatment modalities that are not based on accurate scientific evidence.
The Orthobiologics division was established in 2019 as part of a service line initiative of Cincinnati Sports Medicine and Orthopedic Center- Mercy Health and the Noyes Knee Institute to address the issues of treatment in using Orthobiologics. Under the leadership of knowledgeable and experienced physicians Drs. Frank Noyes, Edward Marcheschi, and Sambhu Choudhury and Brian Chilelli, this division is responsible for every phase of our clinical outcome studies and in particular that these treatment modalities when offered are based on scientific evidence and patient understanding as to their benefits and equally understanding the false claims as patients often seek out these therapies expecting a reparative or curative treatment. As previously stated there is a tremendous amount of information available on the internet, but there is a lack of well designed, prospective clinical outcome studies for knee osteoarthritis. An extensive analysis and systematic review was completed by this Division for treating knee osteoarthritis which formed the basis for the clinical outcome studies initiated. This review reaffirmed the pressing need for accurate clinical information in the treatment algorithm for using these products to treat knee osteoarthritis. Reference is made to the Orthobiologics Treatment section where a summary of current medicines and Orthobiologic approaches is provided.
The first mission of the Orthobiologics Division is the education of patients and medical professionals on the scientific basis and clinical efficacy of recommending Orthobiologic products to treat knee osteoarthritis based on the most current and scientific publications available world-wide. The second mission is to conduct well designed studies on the clinical outcomes and efficacy of selected Orthobiologic products. A final mission is to provide scientifically accurate recommendations to Hospital Systems and patient treatment centers on the incorporation of Orthobiologics into their treatment approaches for knee osteoarthritis.
Current Studies:
- Bone Marrow Aspirate Concentrate (BMAC): Principal Investigator: Dr. Sambhu Choudhury
- Independent analysis of 44 patients. Patient reported outcomes were collected at 3, 6, and 12 months post-operative.
- This robust clinical evaluation will be completed in 2021
- This study specifically examines the role of BMAC in patients who are unable to undergo total knee replacement due to serious medical issues and have disabling pain effecting all activities of daily living. The goal is to determine if a reasonable measure of relief can be obtained to decrease patient pain, allow time for optimization for TKR and to avoid patient turning to opiates to relieve severe knee pain
- Prospective, Randomized Control Trial: Principal Investigators: Dr. Frank Noyes, Dr. Edward Marcheschi
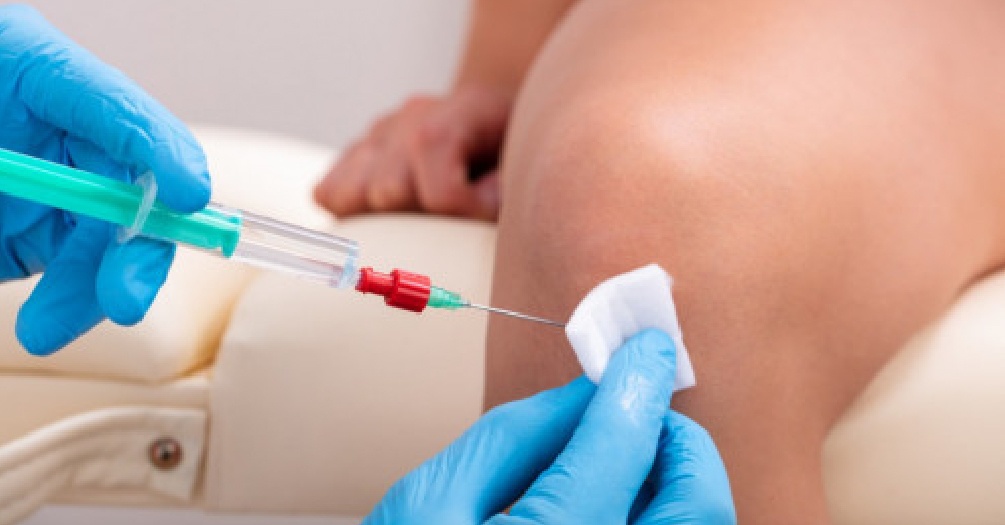
- Prospective RCT of a commercial platelet rich plasma (PRP) intraarticular knee injections based on a treated and control group of 100 patients
- Active patients, age 40-65 will be enrolled into this study.
- Data will be collected at 3, 6, and 12 months post injection. The data collected will be detailed and include subjective, objective, functional assessments and patient reported outcomes using validated knee scores.
- The goal of this study is to make recommendations to Bon Secours – Mercy Health regarding the efficacy of specific PRP products in the Orthopedic service line as well as publication of the results in peer reviewed journals.
- Prospective Randomized Control Trial: Principal Investigators Dr. Brian Chilelli, Dr. Frank Noyes
- This trial will initiate in 2021 and involves a clinical outcome study in using microfragmented adipose tissue for the intra-articular knee injection for osteoarthritis. An RCT is currently in progress and decisions will be made as to conducting the study in this Division or joining a multicenter clinical trial.
When the Best Matters




CSMOC is an award-winning center for orthoapedic treatment in Cincinnati.